
The Sanderson Group Homepage
Chemistry Department
Durham University, Durham, DH1 3LE, UK
Research in the Sanderson group focuses on the interactions of proteins and peptides with membranes.
Research Areas:
- Probing of the reactivity of drugs and other molecules with membrane lipids.
- Understanding the activity of membrane-active peptides and proteins.
- The development of new analytical methods for studying lipids and membranes, including Raman tweezing and linear dichroism spectroscopy.
- The development of model systems for studying the fundamental properties of the interactions between aromatic amino acids and phospholipids.
Recent Publications:
"Lysis of membrane lipids promoted by small organic molecules:
Reactivity depends on structure but not lipophilicity",
Hannah M. Britt, Aruna S. Prakash, Sanna Appleby, Jackie A. Mosely and John M. Sanderson, Sci. Adv., 2020, 6, eaaz8598.
"Far from Inert: Membrane Lipids Possess Intrinsic Reactivity That Has Consequences for Cell Biology",
John M. Sanderson, BioEssays, 2020, e1900147.
"Lytic Reactions of Drugs with Lipid Membranes",
Hannah M. Britt, Clara A. García-Herrero, Paul W. Denny, Jackie A. Mosely and John M. Sanderson, Chem. Sci., 2019, 10, 674–680.
"The Influence of Cholesterol on Melittin Lipidation in Neutral Membranes",
Hannah M. Britt, Jackie A. Mosely and John M. Sanderson, Phys. Chem. Chem. Phys., 2019, 21, 631–640.
"The Association of Defensin HNP-2 with Negatively Charged Membranes: A Combined Fluorescence and Linear Dichroism Study",
Catherine J. Pridmore, Alison Rodger and John M. Sanderson, Biochim. Biophys. Acta - Biomembr., 2016, 1858, 892–903.
Society Membership:
Member of the British Biophysical Society (committee member),
Biophysical Society
and the Royal Society of Chemistry (FRSC).
News
There have been some exciting developments in the research output from the Sanderson group. These centre around the discovery that molecules of all sizes and membrane affinities have the potential to affect the chemistry of membrane lipids. Recent work with small organic molecules have demonstrated a range of activities, from changes in lipid hydrolysis to acyl transfer from the lipid to the molecule.
• Science Advances Article
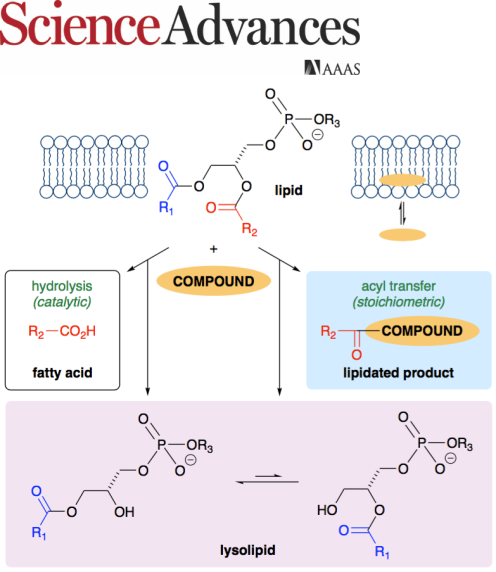 A paper published in Science Advances on 22 April 2020 describes work to investigate the potential for small organic molecules to influence the
stability of membrane lipids. Several small organic molecules are demonstrated to have significant
membrane-lytic potential despite having a low predicted lipophilicity. In aqueous liposome dispersions,
38 aromatic compounds were tested for their ability to either promote lipid hydrolysis or undergo acyl transfer from the lipid to form a lipidated compound.
The notably higher activity for heterocycles such as amino-substituted benzimidazoles and indazoles demonstrates
the potential to predict or "design-in" lytic activity once the rules that govern reactivity are better understood. The nature of
this chemical instability has significant ramifications for the use or presence of lipids in diverse fields such as materials
chemistry, food chemistry, and cell physiology.
To read the article, click here.
A paper published in Science Advances on 22 April 2020 describes work to investigate the potential for small organic molecules to influence the
stability of membrane lipids. Several small organic molecules are demonstrated to have significant
membrane-lytic potential despite having a low predicted lipophilicity. In aqueous liposome dispersions,
38 aromatic compounds were tested for their ability to either promote lipid hydrolysis or undergo acyl transfer from the lipid to form a lipidated compound.
The notably higher activity for heterocycles such as amino-substituted benzimidazoles and indazoles demonstrates
the potential to predict or "design-in" lytic activity once the rules that govern reactivity are better understood. The nature of
this chemical instability has significant ramifications for the use or presence of lipids in diverse fields such as materials
chemistry, food chemistry, and cell physiology.
To read the article, click here.
• Has The Intrinsic Reactivity Of Membrane Lipids Been Overlooked?
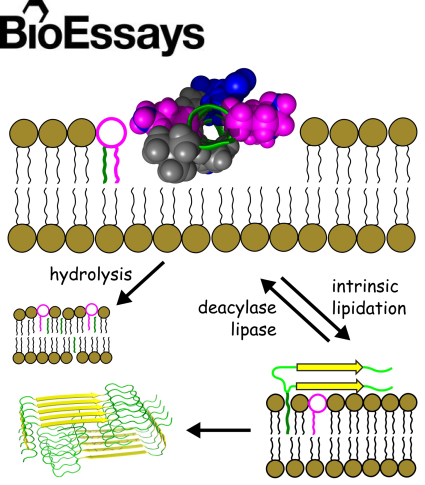 A hypothesis is presented that a fundamental chemical reactivity exists between some non-lipid constituents of cellular membranes and
ester-based lipids, the significance of which is not generally recognised. Many peptides and smaller organic molecules have
now been shown to undergo lipidation reactions in model membranes in circumstances where direct reaction with the lipid is
the only viable route for acyl transfer. Crucially, drugs like propranolol are lipidated in vivo with product profiles that
are comparable to those produced in vitro. Some compounds have also been found to promote lipid hydrolysis. Drugs with high
lytic activity in vivo tend to have higher toxicity in vitro. Deacylases and lipases are proposed as key enzymes that protect
cells against the effects of intrinsic lipidation. The toxic effects of intrinsic lipidation are hypothesized to include a route
by which nucleation can occur during the formation of amyloid fibrils.
To learn more, click here.
A hypothesis is presented that a fundamental chemical reactivity exists between some non-lipid constituents of cellular membranes and
ester-based lipids, the significance of which is not generally recognised. Many peptides and smaller organic molecules have
now been shown to undergo lipidation reactions in model membranes in circumstances where direct reaction with the lipid is
the only viable route for acyl transfer. Crucially, drugs like propranolol are lipidated in vivo with product profiles that
are comparable to those produced in vitro. Some compounds have also been found to promote lipid hydrolysis. Drugs with high
lytic activity in vivo tend to have higher toxicity in vitro. Deacylases and lipases are proposed as key enzymes that protect
cells against the effects of intrinsic lipidation. The toxic effects of intrinsic lipidation are hypothesized to include a route
by which nucleation can occur during the formation of amyloid fibrils.
To learn more, click here.
The Lab
The Sanderson group is located in the Department of Chemistry and the Biophysical Sciences Institute (BSI) at Durham University. To read more about the BSI, click here.



